Use data to understand better
and act better
Transforming complex signals into actionable results requires the right tools and a deep understanding of the clinical context.
We develop signal analysis and processing software designed for research, helping you explore data, test hypotheses, and produce robust results ahead of clinical or regulatory validation.
From sensor to model: end-to-end signal processing expertise
Respiratory physiological signals are complex, sometimes noisy, always rich in information. Interpreting them takes far more than a generic algorithm.
At KerNel Biomedical, we bring solid expertise in analyzing signals from real-world settings, test benches, or clinical sensors. Our approach combines a deep understanding of respiratory challenges with technical mastery of signal processing, from preprocessing to modeling.
We work from data coming from:
- High-fidelity test benches such as the ASL 5000, to reproduce controlled and interpretable conditions.
- Clinical sensors such as oximetry, capnography, or NIRS devices, often used in field studies or applied research.
- Real-life recordings, with their noise, variability, and format constraints.
Our added value is the ability to adapt tools to the nature of the signals, study objectives, and experimental constraints. Each project benefits from a tailored, rigorous analysis, designed to inform scientific or technical decisions rather than just produce numbers.
Software designed for your research needs
Between laborious manual processing and generic solutions that do not fit, we design tools that match your context.
We develop custom software applications designed to streamline respiratory data analysis in research, exploratory validation, or technical clinical studies.
Depending on the project, these tools can:
- Automate large-scale data processing from real or simulated recordings.
- Explore complex physiological indicators and refine their interpretation.
- Test and refine analysis algorithms, to integrate them into a broader validation approach.
Our solutions are designed to integrate into research or scientific structuring projects. They provide a rigorous foundation to explore, demonstrate, and refine your hypotheses, while remaining compatible with your clinical or regulatory validation processes.
Our approach is collaborative, agile, and results-oriented: we help you turn data into practical tools for analysis and decision support.

Concrete cases, varied themes
Our expertise does not stop at algorithms: it is expressed in concrete projects with real clinical or scientific challenges.
We regularly collaborate with research teams, industry, and clinicians on complex respiratory topics, where data processing is central to understanding the phenomena.
The areas we work on include:
- Remote monitoring of ventilated patients or CPAP therapy, with indicator extraction from real ventilatory signals.
- Analysis of patient-ventilator asynchrony, to better characterize the fit between interface, settings, and physiology.
- The study of mask-patient interactions, especially in contexts of leaks, discomfort, or treatment inefficacy.
- Modeling of specific pathologies, such as tracheobronchomalacia, through simulated or real patient profiles.
- Contributions to telemedicine or respiratory rehabilitation, linked to patient quality of life.
These projects illustrate our ability to adapt our tools and methods to current clinical challenges, while meeting rigorous scientific standards.
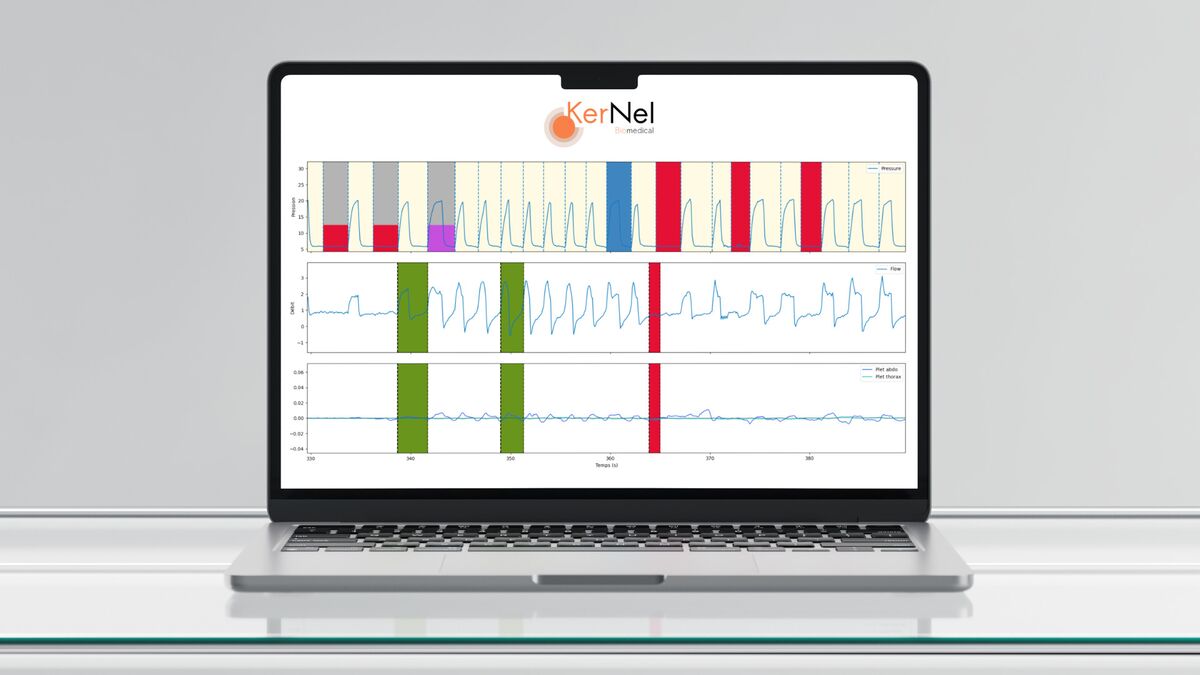
Dotter.science: an online EDC for your clinical studies
Design your eCRFs, enter data, and run your clinical research protocols in a tool built for rigor and collaboration.
Dotter.science is an online platform designed to optimize the design, data entry (eCRF), and electronic management of clinical research protocols (EDC).
The solution helps you to:
- Structure complex eCRFs (phases/steps, form templates, calculated variables).
- Improve data quality with real-time consistency checks and customizable tests.
- Ensure traceability through a complete history of data entry and events (audit trail).
- Simplify data management (Excel/CSV exports, simple or block randomization).
- Work in compliance (GDPR), with hosting on HDS and ISO 27001 certified servers.

Do you need signal processing or data analysis software development?
Book a consultation