
An expert and independent laboratory serving respiratory health.
KerNel Biomedical supports healthcare professionals, researchers, and manufacturers in the design, evaluation, and valorization of medical devices dedicated to respiratory care.
As an independent lab, we combine scientific expertise, technical resources, and a pragmatic approach to produce reliable data, practical training, and decision-support tools.
They trust us
for their academic or industrial projects






Medical device evaluation
High-fidelity test benches to objectively measure your devices' performance.
We conduct technical tests, benchmarks, and regulatory evaluations across a wide range of respiratory support devices: NIV, CPAP, oxygen concentrators, cough assist devices, interfaces, and more.
Our test benches assess performance under realistic and reproducible conditions, to support both R&D and regulatory pathways.
1. Benchmark
Objectively assess your technologies in controlled and realistic conditions, for research, positioning, or communication needs.
2. Performance testing
Produce usable data for regulatory submissions and for certification or CE marking.
3. Advisory
Support the development of robust and compliant devices, aligned with clinical use and field constraints.
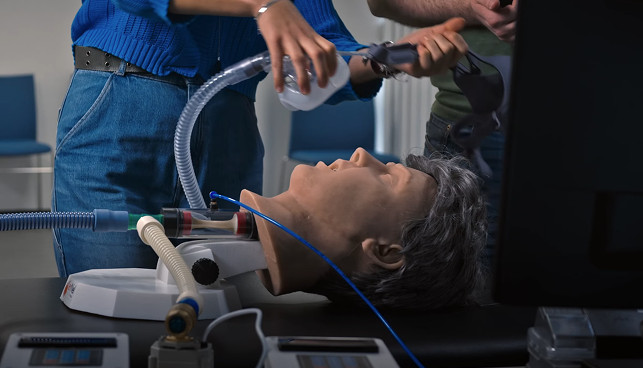
Train for ventilation, act in real-world conditions
Training in ventilation also means learning to use devices in real-world conditions.
Our training, based on simulation with Georges, is designed both for:
- healthcare professionals who want to strengthen practical skills,
- and technicians, caregivers, or assistants facing day-to-day management of a respiratory device.
Advisory and valorization
Give meaning to your data to guide decisions and highlight results.
Beyond measurement, we support you with the interpretation, valorization, and dissemination of your results:
- validation and positioning strategy,
- writing of technical reports or scientific publications,
- support for regulatory or commercial communication.

Software solutions and data processing
Turn respiratory data into insight, from simulation to clinic.
We develop analysis and modeling tools tailored to your research projects, sensors, and experimental protocols. Whether data comes from clinical recordings, real sensors, or test benches simulating controlled conditions, our analyses transform your data into actionable scientific indicators.
Applied research and use cases
Independent, rigorous research focused on clinical impact.
Our scientific work focuses on non-invasive ventilation, pediatrics, pulmonary fibrosis, and resuscitation devices. Each study aims to produce reproducible results that are useful in practice, in collaboration with hospitals, universities, and industry partners.

Make your clinical studies easier
Need to run a clinical study? Dotter.science helps you deliver your research.
Dotter.science is an online platform dedicated to clinical study management, from form design to data centralization and follow-up.
It enables you to structure your protocols, track study progress, and access clean data, ready for analysis, scientific publication, or regulatory use.

Book my demo
We take care of the rest
One priority: help you move forward
From idea to tested, validated, and valued device, KerNel Biomedical supports you at every stage, with a constant demand for scientific rigor and operational efficiency.
Contact us