Medical device evaluation
Producing reliable, rigorous, and interpretable data is essential at every stage of a medical device life cycle.
Whether to convince a procurement committee, position a product on the market, or validate an R&D strategy, robust technical results make the difference. At KerNel Biomedical, we provide an independent lab, a recognized scientific expertise, and the ability to translate performance into clinical use.
Benchmarks to showcase your devices
Your devices have strengths: our test benches make them measurable.
In a competitive sector, clear comparative data is a decisive lever for research, marketing, or product strategy. With custom protocols, we help you position your technologies, support communications, or guide development choices.
- Structured comparison with similar devices, under fair and reproducible conditions.
- Protocols aligned with your objectives, whether for scientific communication (publication, conference) or marketing (promotional materials, sales enablement).
- Simulation of typical or critical clinical cases, to evaluate technical performance in conditions close to medical reality.
- Collaboration with researchers: we also contribute to applied research projects with a constant focus on experimental relevance and reproducibility.
Our independence guarantees total objectivity, for a technical evaluation that is reliable, clear, and defensible.
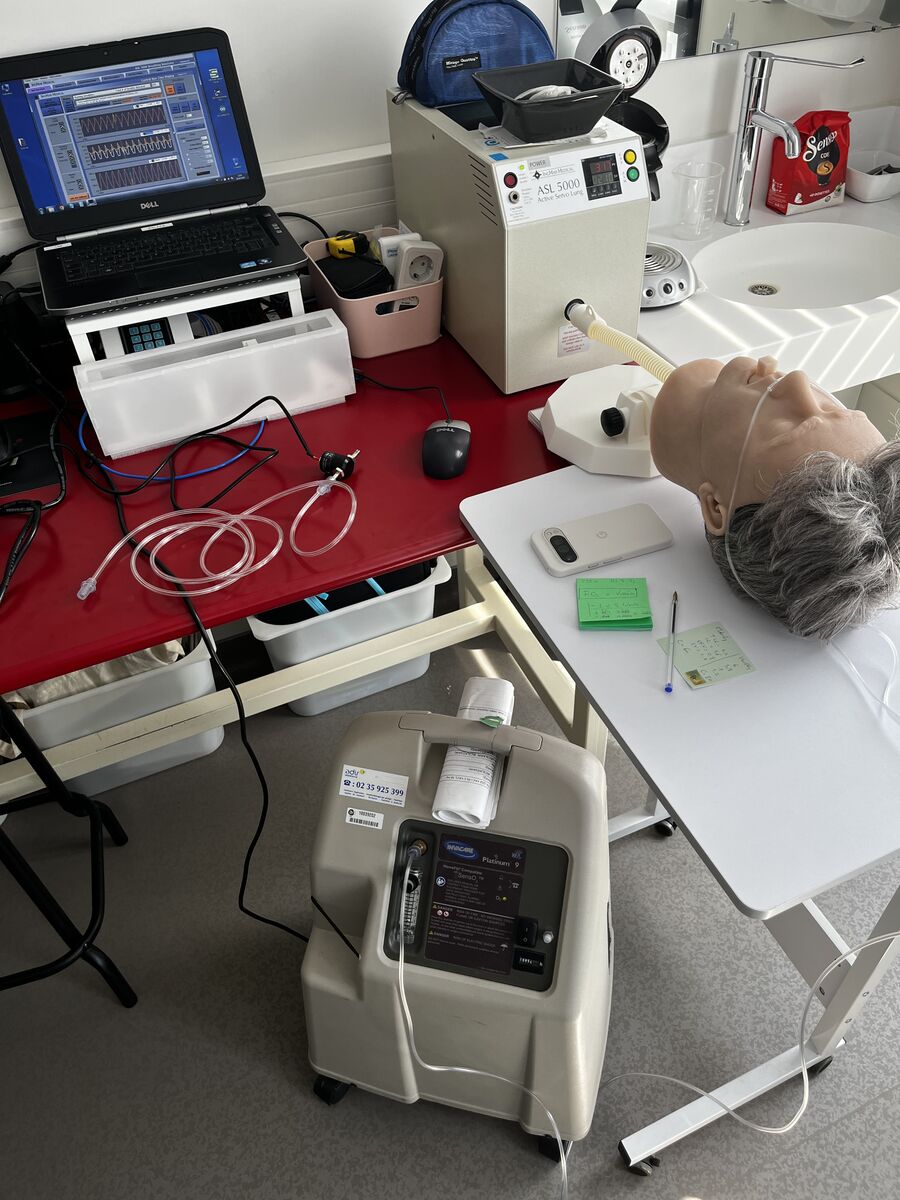
Technical tests for regulatory validation
Certifying a medical device means meeting increasingly precise requirements without compromising timelines or budgets.
Our bench evaluations provide robust, reproducible data to support regulatory submissions accepted by authorities (including CE marking or FDA submissions).
- Realistic protocols, without ethical risks. With our simulation systems, we can reproduce standard clinical conditions and extreme situations such as major leaks, abnormal flows, or severe obstructions. While rare, these events can have a critical clinical impact. These scenarios are difficult to observe in practice and impossible to impose on a patient for ethical reasons, yet they allow you to test device robustness in demanding contexts.
- A complementary or alternative solution to clinical studies. More accessible and faster, bench tests allow for precise characterization of device performance, especially on specific points (e.g., trigger response, effort response, pressure stability).
- A tailored approach aligned with regulatory requirements. We work with your regulatory teams to adapt testing to your submission strategy and authority expectations.
Concrete example: we supported PhysioAssist with specific bench evaluations as part of their regulatory process (FDA 510(k)) in the United States.
- A tool for decision-makers. Our tests also inform purchasing decisions, especially for procurement groups or healthcare providers, by offering objective comparisons between competing devices.
Our objective: provide reliable, usable, and credible technical evidence, within a rigorous, transparent framework aligned with your industrial constraints.

Support for device development
Between the first prototype and the last update, every medical device goes through phases of uncertainty, adjustments, and critical decisions.
Whether designing a new product or optimizing a device already on the market, we support you with an agile, scientific approach aligned with your technical constraints and strategic objectives.
- Iterative, targeted collaboration. We step in at each stage where objective data can guide a decision: algorithm validation, evaluation of a new mode, robustness testing under edge conditions. Each test cycle identifies concrete improvement levers and validates the changes made.
- An accessible lab to test, compare, and adjust. Take advantage of our lab by the day to experiment in controlled conditions. Access our test benches, advanced simulators, and expert support to evolve your products in real time.
- A committed multidisciplinary team. Our engineers, researchers, and clinicians understand your challenges. They speak your language and bring a combined perspective across technical requirements, clinical use, and regulatory expectations.
- Clear data for faster decisions. Instead of waiting for long validations, you get immediately usable results: precise indicators, contextualized and decision-ready.
Our objective: accelerate iterations, make your devices more reliable, and simplify decision-making, with a rigorous method and an experimentation environment that matches your ambitions.

Technical expertise in respiratory support devices
Our core business is the technologies that support breathing.
KerNel Biomedical has recognized expertise across a wide range of medical devices used in ventilation and oxygen therapy, in hospital and at home. We assess performance, robustness, and real-world fit, using simulated scenarios from adults to pediatrics and even newborns.
Key device families we regularly evaluate include:
- Non-invasive ventilation (NIV) : analysis of ventilatory response, synchrony, leak handling, embedded algorithms.
- Continuous positive airway pressure (CPAP) : effectiveness of auto-CPAP modes, detection of respiratory events, leak estimation.
- Nasal high-flow therapy (HFT) : delivered flow accuracy, temperature and hygrometry, and secretion fluidification.
- Oxygen concentrators, continuous or pulsed : gas purity, delivery stability, bolus synchrony, inspired oxygen fraction by pulmonary profile.
- Cough assist devices : secretion mobilization, measurement of generated pressures and flows, effectiveness of insufflation/exsufflation cycles, evaluation of automatic modes.
- Interfaces and accessories : masks, filters, humidifiers, circuits, valves, with the ability to assess leaks, humidification quality, pressure points, and more.
This detailed device knowledge, combined with our test bench expertise, allows us to design protocols tailored to each technology, regardless of development stage or evaluation needs.

Targeted evaluations designed for your devices
We design and carry out targeted technical evaluations that answer your specific questions about device operation and performance.
Here are some examples of services we deliver in client projects:
- Ventilatory performance:
- Flow delivered under simulated effort
- Target pressure accuracy
- Trigger and cycling responsiveness
- Pressure rise time and stability
- Robustness in edge conditions (leaks, obstruction)
- Operating modes:
- Evaluation of auto-adaptation (auto-CPAP, proprietary algorithms)
- Behavior across settings (PEEP, EPAP, S/T mode)
- Response to simulated patient profiles (obstructive, restrictive, pediatric)
- Oxygen delivery analysis:
- Measurement of inspired oxygen fraction (FiO2) based on flow and use
- Evaluation of oxygen purity at concentrator output
- Performance analysis in pulsed or continuous mode
- Sensors and integrated measurements:
- Validation of flow, pressure, humidity, or temperature measurement
- Assessment of displayed value accuracy
- Comparison of exported or transmitted data
- Inter-device comparisons:
- Cross-evaluations across different models
- Neutral benchmarking under standardized conditions
- Tests based on selection criteria for purchasing or decision support
These services can be delivered as part of a full study, a regulatory validation, or a targeted exploratory test. Each protocol is designed specifically to answer your technical question, taking into account usage, measurement, and decision constraints.

Why choose KerNel Biomedical?
You need evidence, not promises.
What sets us apart:
- A unique ability to simulate realistic clinical use on a test bench.
- Total independence, ensuring objectivity.
- A tailored approach that serves your priorities, whether to convince, certify, or improve.
- A constant link between technical performance, real-world use, and compliance requirements.
Do you need your devices tested?
Book a consultation